VIENNA, Austria, July 31, 2023 (ENS) – Heart attacks and strokes arising from atherosclerosis, a disease caused by the accumulation of fats in the inner linings of blood vessels, are responsible for about one-third of all deaths worldwide. In Austria alone last year, 34.7 percent of the total deaths in this country of nine million people were caused by cardiovascular disease.
Researchers focused on atherosclerosis have been seeking the development of new and effective treatments. Now, scientists from the Medical University of Vienna have described a key molecular pathway that offers new fundamental insights into the atherosclerosis progression and may pave the way for new treatment approaches.
The results of their study were published July 26 in the journal “Immunity.” To read it, click here.
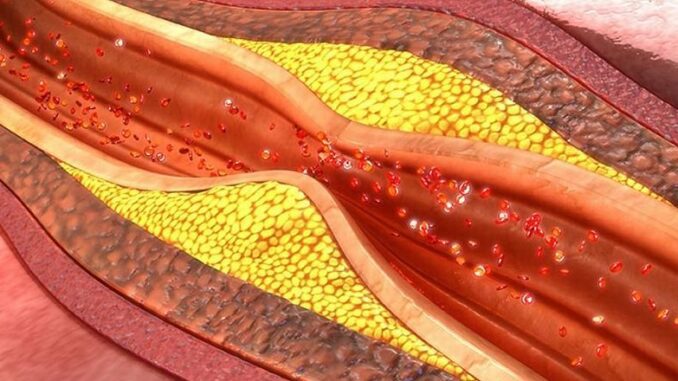
Atherosclerosis is the buildup of fats, cholesterol and other substances in and on the artery walls, a buildup called plaque. This plaque can form atherosclerotic lesions that can cause arteries to narrow, blocking blood flow. The plaque can also burst, leading to a blood clot.
Although atherosclerosis is often considered a heart problem, it can affect any artery in the body from head to toe. Still, atherosclerosis often leads to coronary artery disease, angina, heart attack or heart failure, or stroke.
The Key is in the Complement System
The success of the MedUni Vienna team in identifying a new approach came through their understanding of the complement system.
This system is a group of nearly 60 proteins that are in blood plasma or on the surface of some cells. The proteins work with the host body’s immune system and play a role to protect the body from infections and to remove dead cells and foreign material.
In their study the MedUni Vienna team identified a specific subtype of specialized complement-producing cells called macrophages. These white blood cells are important in the human immune system and that of other vertebrates.
Present in both mouse and human atherosclerotic plaques, macrophages function by engulfing and digesting microorganisms; clearing away debris and dead cells; and stimulating other cells involved in immune function.
But the regulation of cell activity by complement factor H, abbreviated CFH, limits the ability of these macrophages to clear away this of dying cell debris, so more of the debris remains in the bloodstream, aggravating the death of the cells in body tissues such as plaque, a condition known as called plaque necrosis.
Overwhelming necrotic cell death can lead to the instability of atherosclerotic plaques and cardiovascular events, such as heart attacks.
But there is hope among scientists as recent technological advances have led to the discovery of a rich immune cell diversity in atherosclerotic lesions.
These lesions in the arteries are characterized by a profound infiltration of immune cells followed by an accumulation of dying cells that can undergo necrosis if not efficiently handled. The complement system performs critical housekeeping functions by helping macrophages to remove damaged and dying cells.
Introducing C3
Complement component 3, often called C3, is part of this solution. An immune system protein found in the blood, it contributes to our innate immunity.
Part of the complement system is active continuously as it generates “cleavage products of the central component C3 – a process that is highly regulated by complement factor H, or CFH,” the MedUni Vienna scientists said.
Complement activation has been linked to human atherosclerosis before. But the importance of cellular versus systemic complement activation in lesion progression has not been appreciated, the research scientists believe.

The study’s senior author Christoph Binder, in the Department of Laboratory Medicine at MedUni Vienna, said, “There has been increasing evidence that immune cells can also produce a defined set of complement components. However, if and how complement is controlled within these cells has been unknown.”
“We were able to demonstrate that inflammatory monocyte-derived macrophages accumulate complement C3, the central complement component during inflammation, with a concomitant increase in the production of its master regulator, CFH,” Binder explained.
Functional CFH is critical to prevent too much activation of the complement system in the blood.
Using numerous unique mouse models, the researchers dissected the role of CFH in the progression of atherosclerotic lesions.
“First, we made the surprising observation that global lack of CFH displays an overall beneficial impact on plaque progression, which is dependent on its interaction with C3,” lead author Máté Kiss explained.
“Building on these data, we were able pinpoint, that the protective effect is exerted on the cellular level, as selective deletion of CFH in monocytes and macrophages led to a robust decrease in both atherosclerotic lesion size and necrotic area due to an improved capacity to ingest and clear dying cells,” Kiss said.
He explained the importance of this discovery, saying, “This is the first description that highlights the functional importance of a complement regulator to control complement activation within cells with a robust impact on the development of atherosclerosis, and potentially, other chronic inflammatory diseases, too.”
The team’s preclinical findings are matched with data providing clear links to human cardiovascular disease.
Still, the researchers say further investigation is warranted. In the future, they want to focus on “genetic variants in the CFH gene” that “strongly predispose to chronic inflammatory diseases in humans.”
Featured image: Atherosclerotic plaques are masses of oxidized cholesterol, immune cells and dead tissue that form within arterial walls. (Photo from sciencepics/Shutterstock.com courtesy Stanford University)
© 2023, Environment News Service. All rights reserved. Content may be quoted only with proper attribution and a direct link to the original article. Full reproduction is prohibited.

